 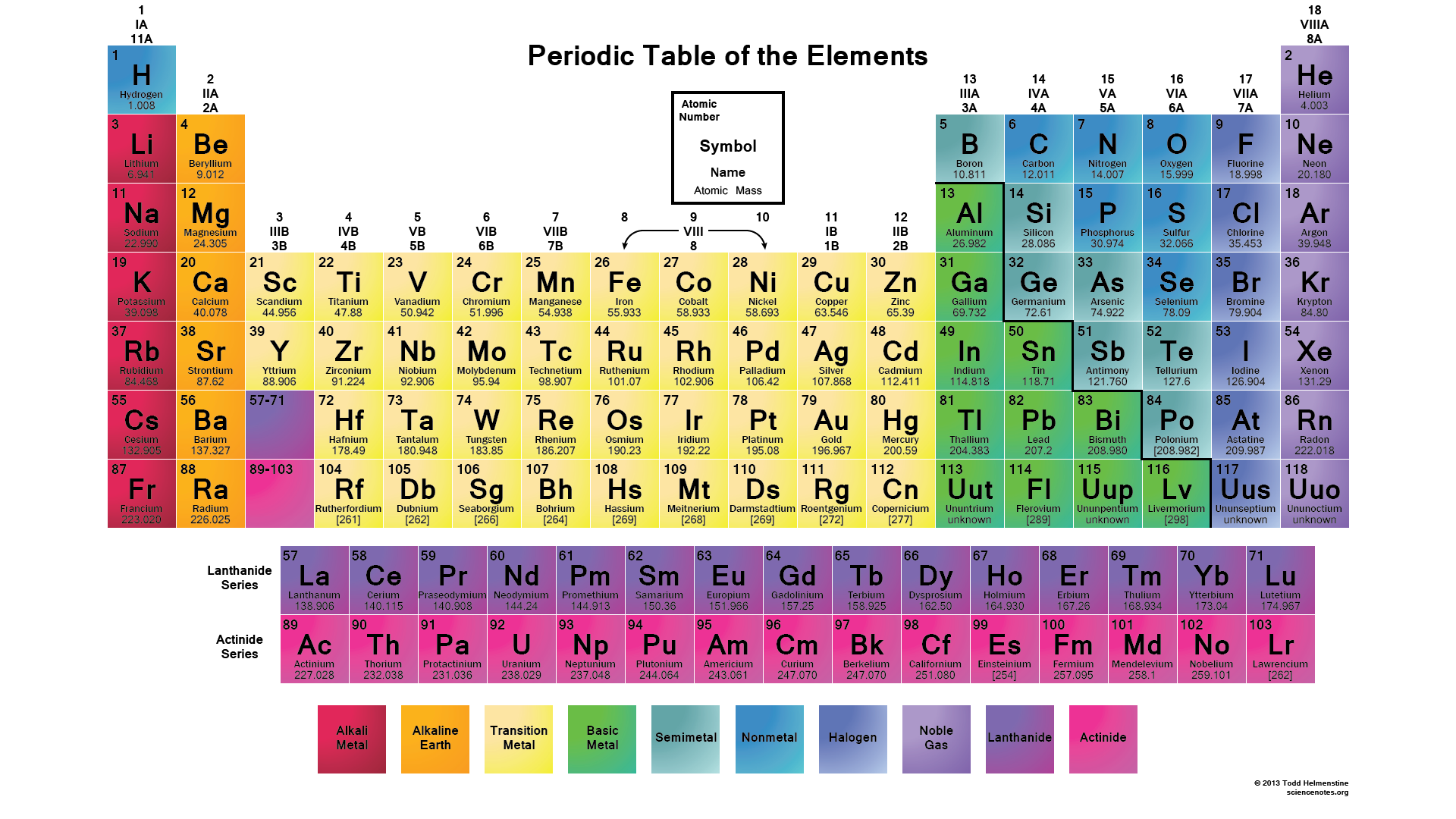
Atomic mass generally increases from left to right across a period, while atomic size generally decreases. The horizontal rows of the periodic table are called periods. 
See also: Alkali metals Atomic number Electron configuration Noble gases Valence 
The position of elements in the periodic table provides a powerful method of classifying not only the physical properties of elements but also their expected properties in molecules and solids. The last column has completely filled valence orbitals leading to chemically inert elements called the noble gases. The first column, known as the alkali metals (albeit with hydrogen, a nonmetal on top), contains elements with just one outer (valence) electron. It is organized such that the vertical columns consist of elements with remarkably similar properties (see illustration). The right side of the periodic table contains the nonmetals (except hydrogen), including halogens (group 17) and noble gases (group 18).Ī list of chemical elements arranged along horizontal rows in increasing atomic number. The left side of the periodic table includes the alkali (group 1) and alkaline earth (group 2) metals, followed by the transition metals in the center. Mendeleev, the periodic table lists every known chemical element in horizontal rows (“periods”) by increasing atomic number.Įach cell in the periodic table contains a symbol representing the element and its atomic number.Įach column of the periodic table contains a family (group) of chemicals with the same number of valence electrons and, therefore, similar chemical properties. Originally developed by the Russian chemist D. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
